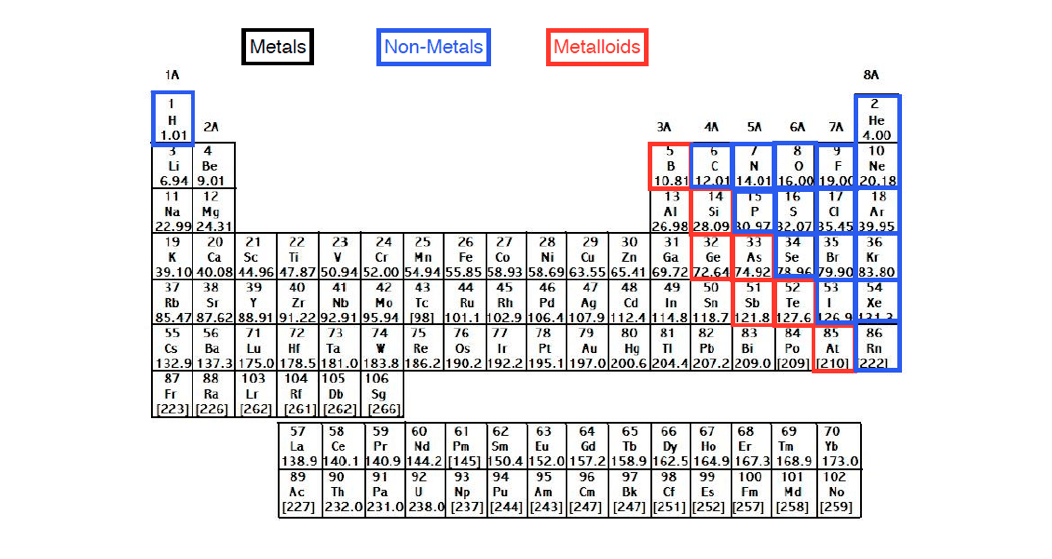
When it comes to medals, medals tend to lose electrons to become positively charged ions called cat ions. So if there number of electron matches one of the nearest noble gasses, now we're going to stay here. 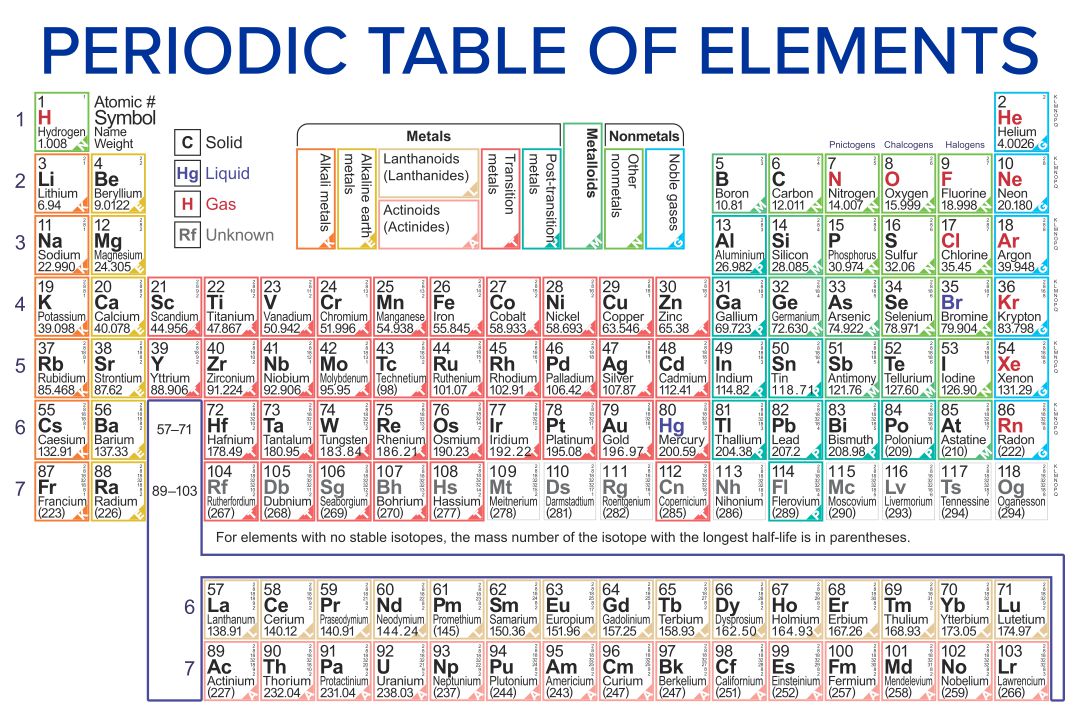
But just realize when it comes to the elements, the leader lose or gain electrons. 
What that means specifically will talk about several chapters from now. The reason these elements want to become the noble gas is because noble gasses themselves have the optimal number off outer shell electrons. Okay, so remember two different ways to describe the same column of the periodic table. Now, remember, your noble gasses are the elements that are found in Group eight A or Group 18. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.Now the elements of the periodic table will either lose or gain electrons to become just like the noble gasses. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Elements Common Charges 1 Charge of Hydrogenionġ+ 2 Charge of Heliumion 0 3 Charge of Lithiumionġ- 10 Charge of Neonion 0 11 Charge of Sodiumionġ- 18 Charge of Argonion 0 19 Charge of PotassiumionĢ+, 3+, 4+, 5+ 24 Charges of Chromiumions 2+, 3+,6+ 25 Charges of Manganeseions 2+, 4+, 7+ 26 Charges of Ironionsģ+ 32 Charges of Germaniumions 4-, 2+, 4+ 33 Charges of Arsenicions 3-, 3+, 5+ 34 Charges of Seleniumions 2-, 4+, 6+ 35 Charges of Bromineionsġ-, 1+, 5+ 36 Charge of Kryptonion 0 37 Charge of Rubidiumion 1+ 38 Charge of Strontiumion 2+ 39 Charge of Yttriumion 3+ 40 Charge of Zirconiumion 4+ 41 Charges of Niobiumions 3+, 5+ 42 Charges of Molybdenumions 3+, 6+ 43 Charge of Technetiumion 6+ 44 Charges of Rutheniumions 3+, 4+, 8+ 45 Charge of Rhodiumion 4+ 46 Charges of Palladiumions 2+, 4+ 47 Charge of Silverionġ+ 48 Charge of Cadmiumion 2+ 49 Charge of Indiumion 3+ 50 Charges of Tinions 2+, 4+ 51 Charges of Antimonyions 3-, 3+, 5+ 52 Charges of Telluriumions 2-, 4+, 6+ 53 Charge of Iodineionġ- 54 Charge of Xenonion 0 55 Charge of Cesiumionġ+ 56 Charge of Bariumion 2+ 57 Charge of Lanthanumion 3+ 58 Charges of Ceriumions 3+, 4+ 59 Charge of Praseodymiumion 3+ 60 Charges of Neodymiumions 3+, 4+ 61 Charge of Promethiumion 3+ 62 Charge of Samariumion 3+ 63 Charge of Europiumion 3+ 64 Charge of Gadoliniumion 3+ 65 Charges of Terbiumions 3+, 4+ 66 Charge of Dysprosiumion 3+ 67 Charge of Holmiumion 3+ 68 Charge of Erbiumion 3+ 69 Charge of Thuliumion 3+ 70 Charge of Ytterbiumion 3+ 71 Charge of Lutetiumion 3+ 72 Charge of Hafniumion 4+ 73 Charge of Tantalumion 5+ 74 Charge of Tungstenion 6+ 75 Charges of Rheniumions 2+, 4+, 6+, 7+ 76 Charges of Osmiumions 3+, 4+, 6+, 8+ 77 Charges of Iridiumions 3+, 4+, 6+ 78 Charges of Platinumions 2+, 4+, 6+ 79 Charges of Goldions 1+, 2+, 3+ 80 Charges of Mercuryions 1+, 2+ 81 Charges of Thalliumions 1+, 3+ 82 Charges of Leadions 2+, 4+ 83 Charge of Bismuthion 3+ 84 Charges of Poloniumions 2+, 4+ 85 Charge of Astatineion Unknown 86 Charge of Radonion 0 87 Charge of Franciumion Unknown 88 Charge of Radiumion 2+ 89 Charge of Actiniumion 3+ 90 Charge of Thoriumion 4+ 91 Charge of Protactiniumion 5+ 92 Charges of Uranium ions 3+, 4+, 6+ Free Gift for you: Interactive Periodic Table 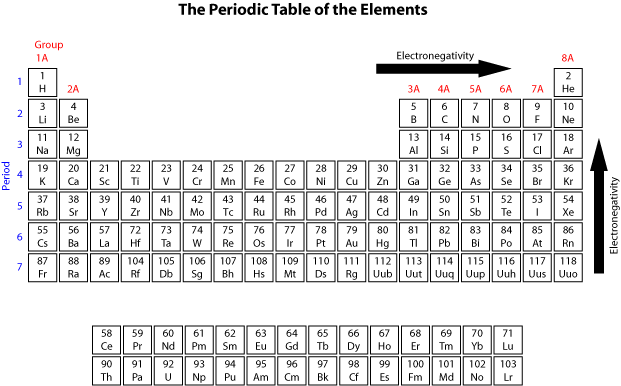
List of elements with their common ionic charges are mentioned below.Įlements with multiple ionic charges are also mentioned in this table. When atoms gain electron/s, the negatively charged ion is formed, and when the atoms lose electron/s, the positively charged ion is formed. This electric charge generated on the ion is known as Ionic charge. Ionic charge: When the atom loses or gains one or more electrons, the electric charge is generated (and an ion is formed).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
